Premium Information is available for this item - Upgrade for $1 a day
6530-01-302-0645
Sterilization Envelope
6530013020645 013020645 92308
An envelope usually made of paper or plastic material which permits steam and heat penetration and the exhaustion of air in the sterilization process. It may be self or heat sealed. It is used in the sterilization of syringes, needles, small surgical instruments, pipettes and the like. Excludes BAG, STERILIZATION; BAG, STERILIZATION-BIOHAZARD DISPOSAL; and WRAPPER, STERILIZATION. View more Sterilization Envelope
![]()
January 2023
4
 Marketplace 6530-01-302-0645
Marketplace 6530-01-302-0645
Request a Quotation from participating marketplace vendors
 Related Documents 6530-01-302-0645 5+ Documents (More...)
Related Documents 6530-01-302-0645 5+ Documents (More...)
6530-01-302-0645 5+ Documents ( More... ) https//www.nsnlookup.com /fsg-65/fsc-6530/us 6530-01-302-0645
Day 6530-01-302-0645 RQST NE Related Documents 6530-01-302-0645 5+ Documents ( More... ) https//www.nsnlookup.com
0029 6530-12-277-0039 /fsc-6530/de 6530-12 https https//www.nsnlookup.com /fsg-65/fsc-6530/us 6530-01-302-0645
Sterilization Envelope 6530013020645,92308 372-6358 6530-01-300-0910 6530-01-302-0645 https https
... ) https//www.nsnlookup.com https//www.nsnlookup.com /fsg-65/fsc-6530/de 6530-12-372-6358 6530-01-302-0645
0029 6530-12-372-6358 https https//www.nsnlookup.com / www.nsnlookup.com /fsg-65/fsc-6530/us 6530-01-302-0645
6530-01-500-4944 6530-01-004-3468 6530-00-006-5948 https//www.nsnlookup.com /fsg-65/fsc-6530/us 6530-01-302-0645
Sterilization Envelope 6530013020645,92308 6530-12-372-6358 6530-01-300-0910 6530-01-302-0645 https
www.nsnlookup.com /fsg-65/fsc-6530/us 6530-01-500-4944 Sterilization Envelope 6530015004944,02-1469 300-0910 6530-01-302-0645
6530-01-500-4944 6530 https https//www.nsnlookup.com /fsg-65/fsc-6530/us 6530-01-302-0645 Sterilization
 Restrictions 6530-01-302-0645
Restrictions 6530-01-302-0645
6530-01-302-0645 is a Sterilization Envelope that does not have a nuclear hardened feature or any other critical feature such as tolerance, fit restriction or application. This item is considered a low risk when released from the control of the Department of Defense. The item may still be subject to the requirements of the Export Administration Regulations (EAR) and the Code of Federal Regulations (CFR). This item is not suspected to be hazardous. This item does not contain a precious metal.
 End Users 6530-01-302-0645
End Users 6530-01-302-0645
- MOE Rule:
- V540
- Effective Date:
- 1 Apr 1991
 Approved Sources 6530-01-302-0645
Approved Sources 6530-01-302-0645
- Part Number
- Manufacturer
- Status
- 92308
- Manufacturer
- 19484 - Baxter Healthcare Corp (Obsolete)
- Primary Buy
- Primary Buy
 Datasheet 6530-01-302-0645
Datasheet 6530-01-302-0645
- Characteristic
- Specifications
- FIIG
- Specifications
- A11100
- Assembly Method [AFPN]
- Heat Seal
- Opening Location [ARQT]
- Ends
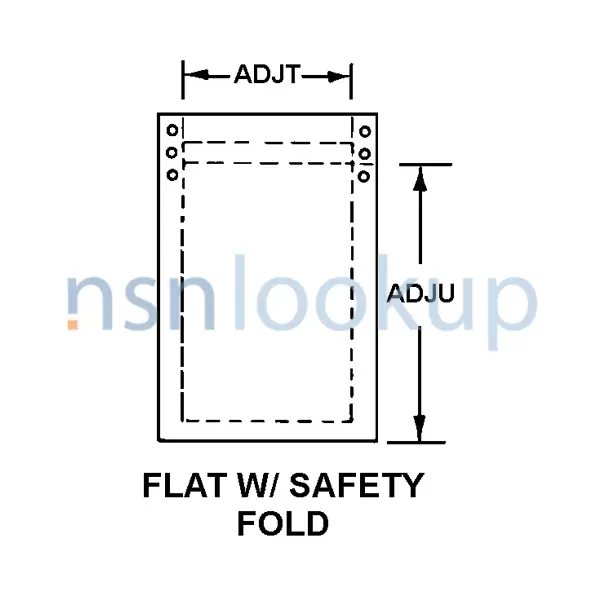
- Style Designator [STYL]
- Flat W/Safety Fold
- Inside Length [ADJU]
- 8.000 Inches Nominal
- Inside Width [ADJT]
- 3.500 Inches Nominal
- Material And Location [ANNQ]
- Paper Single Wall And Plastic Single Wall
 Similar Supply Items to 6530-01-302-0645
Similar Supply Items to 6530-01-302-0645

















 Freight Information 6530-01-302-0645
Freight Information 6530-01-302-0645
6530-01-302-0645 has freight characteristics.. 6530-01-302-0645 has a variance between NMFC and UFC when transported by rail and the description should be consulted.