Premium Information is available for this item - Upgrade for $1 a day
6515-01-624-2620
Intravenous Catheter And Needle Unit
6515016242620 016242620 TPAK 8304817 IN/123
An item consisting of a cannulated needle with an intravenous catheter, and it may contain additional components, such as a hypodermic syringe, needle guard, flow control plug, and the like. The catheter is inserted into the vein by way of the needle's puncture, and is left in position for continued intravenous administration after the needle is withdrawn. A syringe, when supplied, is used to better control insertion of the catheter needle unit or for blood aspiration. View more Intravenous Catheter And Needle Unit
![]()
September 2023
4
 Product Information 6515-01-624-2620
Product Information 6515-01-624-2620
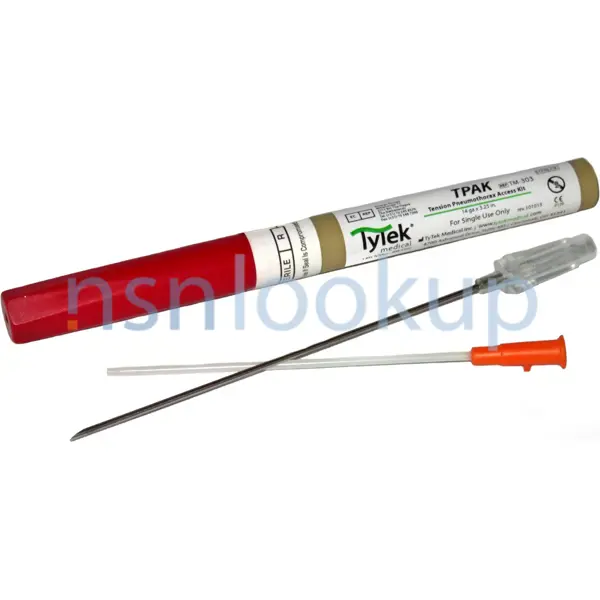
The TyTek Tension Pneumothorax Access Pack (TPAK) is a 14 gauge by 3.25-inch needle and catheter for use in the management of combat casualties who present with the signs and symptoms of a tension pneumothorax.
 Marketplace 6515-01-624-2620
Marketplace 6515-01-624-2620
Request a Quotation from participating marketplace vendors
 Related Documents 6515-01-624-2620 5+ Documents (More...)
Related Documents 6515-01-624-2620 5+ Documents (More...)
Intravenous ,TPAK Fluke TPAK /fsg TPAK /fsg-65/fsc-6515/us 6515 /fsg-65/fsc-6515/us 6515-01-624
us 6515-01-624-2620 Intravenous Catheter And Needle Unit,TPAKTPAK Supply TPAK www.nsnlookup.com https
TPAK Fluke TPAK Fluke TPAK Supply TPAK www.nsnlookup.com /fsg-66/fsc-6625/it ,TPAK 6625- 015579652,TPAK
Fluke TPAK Fluke https // TPAK Fluke Fluke TPAK Fluke TPAK Fluke TPAK ,TPAK https//www.nsnlookup.com
537-4094 6515-01-419-6393 6515-01-273-8625 6515-00-458-8414 6515-01-548-1027 6515-01-530-0782 6515-01-624-2620
-6515/tr 6515-27-019-5299 Surgical Absorbable Suture 6515270195299 548-1027 6515-01-530-0782 6515-01-624-2620
www.nsnlookup.com /fsg-65/fsc-6515/us 6515-01-425-5175 Tissue Forceps 6515014255175 014255175 530-0782 6515-01-624-2620
6625- 015579652,TPAK Fluke TPAK Fluke https//www.nsnlookup.com Fluke TPAK Fluke TPAK https//www.nsnlookup.com
/fsg-65/fsc-6515/us 6515-01-624-2620 Intravenous Catheter And Needle Unit,TPAK TPAK Supply TPAK www.nsnlookup.com
 Restrictions 6515-01-624-2620
Restrictions 6515-01-624-2620
6515-01-624-2620 is a Intravenous Catheter And Needle Unit that does not have a nuclear hardened feature or any other critical feature such as tolerance, fit restriction or application. Demilitarization of this item has been confirmed and is not currently subject to changes. This item is considered a low risk when released from the control of the Department of Defense. The item may still be subject to the requirements of the Export Administration Regulations (EAR) and the Code of Federal Regulations (CFR). This item is not suspected to be hazardous. This item does not contain a precious metal.
 Import and Export 6515-01-624-2620
Import and Export 6515-01-624-2620
- Schedule B
- Subscribe to View Schedule B
- HTS Code
- Subscribe to View HTS Code
 End Users 6515-01-624-2620
End Users 6515-01-624-2620
- Australia (ZA01)
- Effective Date:
- 1 Dec 2013
 Approved Sources 6515-01-624-2620
Approved Sources 6515-01-624-2620
- Part Number
- Manufacturer
- Status
- TPAK
- Manufacturer
- 6TL45 - Tytek Medical Inc (Active)
- Primary Buy
- Primary Buy
- 8304817
- ZBF76 - Anspec Pty Ltd (Active)
- Secondary Buy
- Secondary Buy
- IN/123
- Z0RT3 - Sp Services (Aus) Llp (Active)
- Secondary Buy
- Secondary Buy
 Datasheet 6515-01-624-2620
Datasheet 6515-01-624-2620
- Characteristic
- Specifications
- FIIG
- Specifications
- A22100
- Part Name Assigned By Controlling Agency [CXCY]
- Tpak-Chest Decompression Needle
- Special Features [FEAT]
- Strong Reliable Tension Pneumothorax Needle Allows For Secure Placement Of Cathether For Continuous Relief During Thoracostomy; Rugged Tube With Textured Easy Open Ihex Cap; Capless Flash Chamber For Instant Confirmation Of Needle Placement; Convenient, Compact Size For Maximum Portability; Shock Resistant Precision Dimensioned Case And Needle Hub Ensure That Chest Decompression Neele And Cathether Secure Upon Deployment For Safety
 Similar Supply Items to 6515-01-624-2620
Similar Supply Items to 6515-01-624-2620

















 Freight Information 6515-01-624-2620
Freight Information 6515-01-624-2620
6515-01-624-2620 has freight characteristics managed by Australia.. 6515-01-624-2620 has a variance between NMFC and UFC when transported by rail and the description should be consulted.