Premium Information is available for this item - Upgrade for $1 a day
6515-01-421-0881
Percutaneous Entry Needle
6515014210881 014210881 55018
A sterile, disposable item designed to be used for arterial and venous puncture procedures. View more Percutaneous Entry Needle
![]()
June 2023
5
 Marketplace 6515-01-421-0881
Marketplace 6515-01-421-0881
Request a Quotation from participating marketplace vendors
 Related Documents 6515-01-421-0881 5+ Documents (More...)
Related Documents 6515-01-421-0881 5+ Documents (More...)
-01-421-0881 Percutaneous Entry Needle 6515014210881 RQST NE Updated Every Day 6515-01-421-0881 RQST
Updated Every Day 6515-01-421-0881 RQST NE Related -01-421-0881 Percutaneous Entry Needle 6515014210881
Percutaneous Entry Needle 6515014210881 fsc-6515/us 6515-01-420-7245 Needle 6515014210881 https
6515015038811 Percutaneous 6515014210881 https/ fsg-65/fsc-6515/us 6515-01-503- https//www.nsnlookup.com
Percutaneous Entry Needle 6515014210881 Percutaneous Entry Needle 6515014210881 https/ https//www.nsnlookup.com
Needle 6515270441281 RQST NE Updated Every Day https//www.nsnlookup.com Percutaneous Entry Needle 6515014210881
Percutaneous Entry Needle 6515014210881 Percutaneous Entry Needle 6515015038811 us 6515 https//
503-8811 Percutaneous Entry Needle 6515015038811 Percutaneous Entry Needle 6515270441281 Needle 6515014210881
Percutaneous Entry Needle 6515014210881 Entry Needle 6515014210881 1281 6515-01 -421-0881 https
421-0077 6515-01-362-8297 6515-22-608-0815 6515-99-898-3200 6515-01-440-9559 6515-27-044-1281 6515-01-421-0881
 Restrictions 6515-01-421-0881
Restrictions 6515-01-421-0881
6515-01-421-0881 is a Percutaneous Entry Needle that does not have a nuclear hardened feature or any other critical feature such as tolerance, fit restriction or application. Demilitarization of this item has been confirmed and is not currently subject to changes. This item is considered a low risk when released from the control of the Department of Defense. The item may still be subject to the requirements of the Export Administration Regulations (EAR) and the Code of Federal Regulations (CFR). This item is not suspected to be hazardous. This item does not contain a precious metal.
 Import and Export 6515-01-421-0881
Import and Export 6515-01-421-0881
- Schedule B
- Subscribe to View Schedule B
- HTS Code
- Subscribe to View HTS Code
 End Users 6515-01-421-0881
End Users 6515-01-421-0881
- MOE Rule:
- V540
- Effective Date:
- 1 Nov 1995
 Approved Sources 6515-01-421-0881
Approved Sources 6515-01-421-0881
- Part Number
- Manufacturer
- Status
- 55018
- Manufacturer
- 88009 - U S Dept Of Veterans Affairs (Active)
- Primary Buy
- Primary Buy
 Datasheet 6515-01-421-0881
Datasheet 6515-01-421-0881
- Characteristic
- Specifications
- FIIG
- Specifications
- A21800
- Design Designation [ANEH]
- Flashback
- Style Designator [STYL]
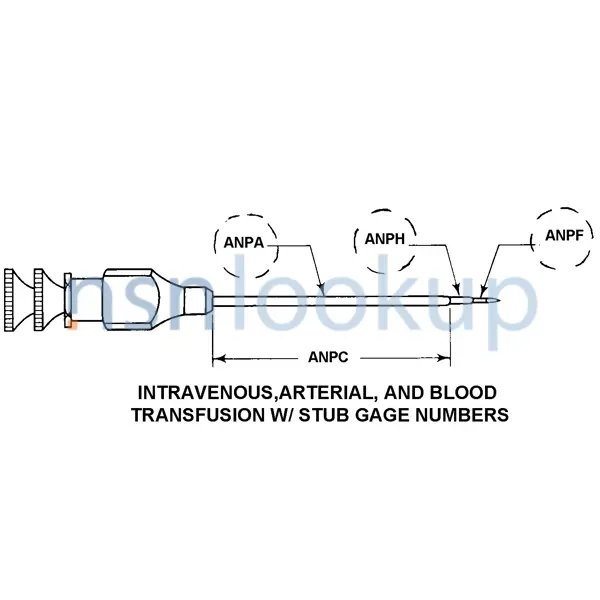
- Intravenous, Arterial, And Blood Transfusion W/ Stub Needle Gage Number(S)
- Cannula Outside Diameter [ANPA]
- 1.200 Millimeters Nominal
- Cannula Length [ANPC]
- 2.750 Inches Nominal
- Material And Location [ANNQ]
- Any Acceptable Overall
- Unit Package Quantity [AGUC]
- 20
- Features Provided [CBBL]
- Disposable
- Special Features [FEAT]
- 18 Gage; Easy-Grip Hub; Porous Plug; Used For Anterior Single-Wall Punctures.
 Similar Supply Items to 6515-01-421-0881
Similar Supply Items to 6515-01-421-0881


















 Freight Information 6515-01-421-0881
Freight Information 6515-01-421-0881
6515-01-421-0881 has freight characteristics.. 6515-01-421-0881 has a variance between NMFC and UFC when transported by rail and the description should be consulted.