Premium Information is available for this item - Upgrade for $1 a day
6515-01-391-9709
Surgical Nonabsorbable Suture
6515013919709 013919709 X935H ETHIBOND
An item designed for stitching and thus securing tissues. It is designed of a material which cannot be dissolved and absorbed in such tissues. View more Surgical Nonabsorbable Suture
![]()
January 2023
2

 Marketplace 6515-01-391-9709
Marketplace 6515-01-391-9709
Request a Quotation from participating marketplace vendors
 Related Documents 6515-01-391-9709 5+ Documents (More...)
Related Documents 6515-01-391-9709 5+ Documents (More...)
your Account Upgrade 6515-01-392-0189Surgical Nonabsorbable Suture6515013920189 013920189 X844H ETHIBOND
your Account Upgrade 6515-01-392-0189Surgical Nonabsorbable Suture6515013920189 013920189 X844H ETHIBOND
your Account Upgrade 6515-01-392-0189Surgical Nonabsorbable Suture6515013920189 013920189 X844H ETHIBOND
your Account Upgrade 6515-01-392-0181Surgical Nonabsorbable Suture6515013920181 013920181 X833H ETHIBOND
your Account Upgrade 6515-01-356-3830Surgical Nonabsorbable Suture6515013563830 013563830 CX45D ETHIBOND
your Account Upgrade 6515-01-392-0189Surgical Nonabsorbable Suture6515013920189 013920189 X844H ETHIBOND
Surgical Nonabsorbable Suture 6515013919709 6515-01-391-9709 6515013919709 013919709 is an item
Nonabsorbable Suture6515013563830 013563830 CX45D ETHIBOND your Account Upgrade 6515-01-356-3830Surgical
Nonabsorbable Suture6515013563830 013563830 CX45D ETHIBOND your Account Upgrade 6515-01-356-3830Surgical
 Restrictions 6515-01-391-9709
Restrictions 6515-01-391-9709
6515-01-391-9709 is a Surgical Nonabsorbable Suture that does not have a nuclear hardened feature or any other critical feature such as tolerance, fit restriction or application. This item is considered a low risk when released from the control of the Department of Defense. The item may still be subject to the requirements of the Export Administration Regulations (EAR) and the Code of Federal Regulations (CFR). This item is not suspected to be hazardous. This item does not contain a precious metal.
 Import and Export 6515-01-391-9709
Import and Export 6515-01-391-9709
- Schedule B
- Subscribe to View Schedule B
- HTS Code
- Subscribe to View HTS Code
 End Users 6515-01-391-9709
End Users 6515-01-391-9709
- MOE Rule:
- V542
- Effective Date:
- 1 Jun 1994
 Approved Sources 6515-01-391-9709
Approved Sources 6515-01-391-9709
- Part Number
- Manufacturer
- Status
- X935H
- Manufacturer
- 10614 - Ethicon, Inc (Obsolete)
- Original Design
- Original Design
- ETHIBOND
- 10614 - Ethicon, Inc (Obsolete)
- Canceled/Obsolete
- Canceled/Obsolete
 Datasheet 6515-01-391-9709
Datasheet 6515-01-391-9709
- Characteristic
- Specifications
- FIIG
- Specifications
- A21900
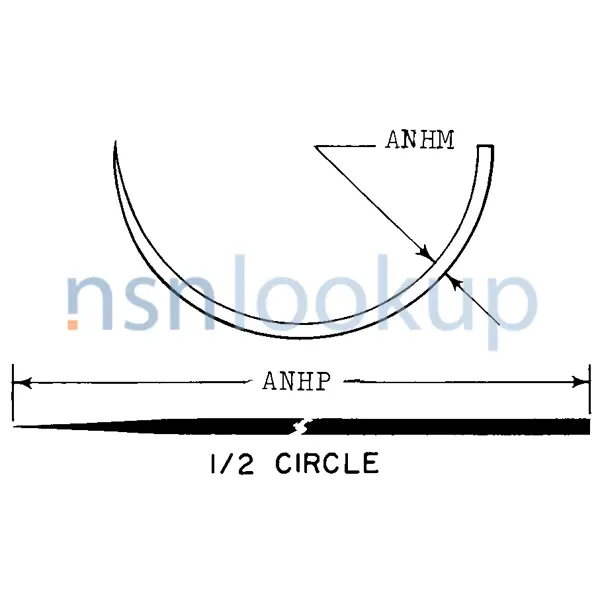
- Armed Suture Needle Point Style [APRH]
- Tapercut
- Armed Suture Needle Style [APWL]
- 1/2 Circle
- Color [HUES]
- Green
- End Type [APTD]
- Double Armed
- Needle Design Designation [APSH]
- Cardiovascular
- Immediate Container Quantity [ANNX]
- 36
- Immediate Container Type [ANNW]
- Packet, Foil, Sealed
- Length [ABRY]
- 750.0 Millimeters Nominal
- Material And Location [ANNQ]
- Steel, Corrosion Resisting Needle And Plastic, Polyester Suture
- Quantity Within Each Immediate Container [ANNY]
- 1
- Size Designator [AJXE]
- 4-0
- Special Features [FEAT]
- Each Suture Packaged With 2 Atraloc V-5 Needles
- Unit Package Quantity [AGUC]
- 36
- Sterility [AKMX]
- Sterile
- Strand Fiber Arrangement [APZJ]
- Braided
 Management Data 6515-01-391-9709
Management Data 6515-01-391-9709
- Effective Date
- Organization
- Unit of Issue
- Unit Price
- Qty Unit Pack
- Unit of Issue
- Jun 1994
- Veterans Administration (VA)
- BX
- Subscribe
- BX
- MOE
- USC
- Code
- Statement
- Order of Use
- Jump To Code
- Qty Per Assy
- UOM
- Technical Document
- Quantative Expression
- Code
- Veterans Administration (VA)
- V
- K
- U/I Contains 36 Ea
- 00000000036EA
- K
 NATO Stock Numbers Related to 6515-01-391-9709
NATO Stock Numbers Related to 6515-01-391-9709
 Freight Information 6515-01-391-9709
Freight Information 6515-01-391-9709
6515-01-391-9709 has freight characteristics.. 6515-01-391-9709 has a variance between NMFC and UFC when transported by rail and the description should be consulted.