Premium Information is available for this item - Upgrade for $1 a day
6515-01-354-4312
Suture Needle
6515013544312 013544312 1824-3
A surgical item designed for use with a suture to transfix and stitch tissue of wounds and blood vessels. View more Suture Needle
![]()
January 2023
4
 Marketplace 6515-01-354-4312
Marketplace 6515-01-354-4312
Request a Quotation from participating marketplace vendors
 Related Documents 6515-01-354-4312 5+ Documents (More...)
Related Documents 6515-01-354-4312 5+ Documents (More...)
 Restrictions 6515-01-354-4312
Restrictions 6515-01-354-4312
6515-01-354-4312 is a Suture Needle that does not have a nuclear hardened feature or any other critical feature such as tolerance, fit restriction or application. This item is considered a low risk when released from the control of the Department of Defense. The item may still be subject to the requirements of the Export Administration Regulations (EAR) and the Code of Federal Regulations (CFR). This item is not suspected to be hazardous. The precious metals content of this item is unknown.
 Import and Export 6515-01-354-4312
Import and Export 6515-01-354-4312
- Schedule B
- Subscribe to View Schedule B
- HTS Code
- Subscribe to View HTS Code
 End Users 6515-01-354-4312
End Users 6515-01-354-4312
- NASA (G860)
- Effective Date:
- 1 Apr 1991
 Approved Sources 6515-01-354-4312
Approved Sources 6515-01-354-4312
- Part Number
- Manufacturer
- Status
- 1824-3
- Manufacturer
- 18521 - Anchor Products Company (Obsolete)
- Original Design
- Original Design
 Datasheet 6515-01-354-4312
Datasheet 6515-01-354-4312
- Characteristic
- Specifications
- FIIG
- Specifications
- A21900
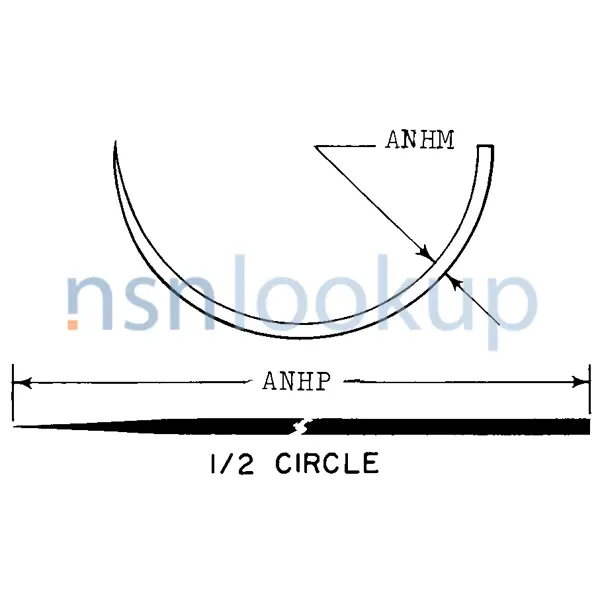
- Needle Design Designation [APSH]
- Mayo Catgut Or Mayo Intestinal
- Immediate Container Quantity [ANNX]
- 12
- Immediate Container Type [ANNW]
- Package
- Special Features [FEAT]
- Size 3
- Sterility [AKMX]
- Nonsterile
- Style Designator [STYL]
- 1/2 Circle
 NATO Stock Numbers Related to 6515-01-354-4312
NATO Stock Numbers Related to 6515-01-354-4312
 Freight Information 6515-01-354-4312
Freight Information 6515-01-354-4312
6515-01-354-4312 has freight characteristics.. 6515-01-354-4312 has a variance between NMFC and UFC when transported by rail and the description should be consulted.